Understanding the Purification Power of Reverse Osmosis: What It Removes and Why It Matters
Facilities and large-scale operations should consider implementing reverse osmosis systems due to their unparalleled efficiency in purifying water, which is crucial for meeting stringent water quality standards and protecting critical equipment from scale and contamination.
Reverse osmosis (RO) is a pressure-driven membrane separation process that utilizes a semipermeable membrane to remove dissolved solids, organics, proteins, particles, and other contaminants in water. First developed in the 1950s for desalination applications, reverse osmosis has since expanded into a variety of industrial, commercial, and residential settings. Reverse osmosis is now used for drinking water production from seawater or brackish sources. It’s also used for wastewater concentration and recycling, food and beverage processing, pharmaceutical purification, boiler feed water treatment, and microelectronics fabrication.
Compared to other separation processes, like filtration or distillation, reverse osmosis offers superior contaminant removal capabilities through a scalable and efficient process. This article will dive into the purification process of reverse osmosis and why it matters.
How Reverse Osmosis Works
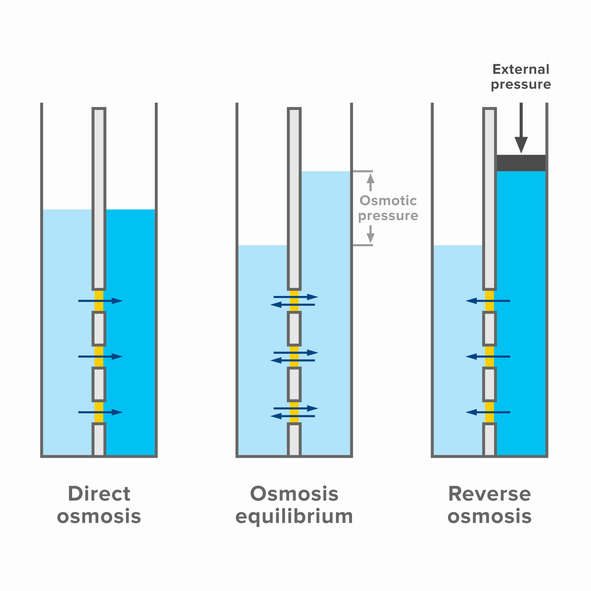
Reverse osmosis utilizes a semipermeable membrane that allows water to pass through while rejecting most dissolved solutes and suspended particulates. An applied pressure must exceed the inherent osmotic pressure of the feed solution to preferentially drive water across the membrane, leaving concentrated draw solution behind (hence the name reverse osmosis). The membrane acts as a selective barrier due to its extremely tiny pore sizes of 0.0001 to 0.001 microns.
Separation by reverse osmosis membranes is governed by size exclusion or steric hindrance principles. Ions or molecules with sizes approaching the membrane’s pore dimensions are repelled, while smaller water molecules pass through largely unimpeded. As such, selectivity depends heavily on properties like molecular weight or effective solute size relative to membrane pore sizes.
The molecular weight cut off (MWCO) rating of a membrane describes the molecular weight of a solute that is 90% rejected. As MWCO decreases, the ability to remove smaller compounds improves. Solutes are concentrated in the leftover brine solution, while high-purity water permeates through the membrane.
Comparison with Other Filtration Technologies
Reverse osmosis is often compared to other pressure-driven membrane filtration processes like microfiltration, ultrafiltration, and nanofiltration in terms of separation size and capabilities. Microfiltration is ideal for removing large particles and bacteria. Nanofiltration shares similarities with RO but generally has larger pore sizes, making it less effective against salts but suitable for removing organics and multivalent ions.
Ultrafiltration is effective against larger molecules and particles, often serving as a pre-treatment step to protect RO membranes. So while overlapping in some size ranges, RO sits on the smallest end of membrane filtration. It should also be mentioned that since RO does not reject 100% of all contaminants, additional “polishing” filters can be added after filtration, such as ion exchange resins or activated carbon.
Contaminant Removal Capabilities
Understanding what reverse osmosis removes can help industrial users understand what applications the process should be used for. So, what does reverse osmosis remove?
Dissolved Inorganics
Reverse osmosis effectively rejects dissolved inorganic salts, minerals, and metals from water through size exclusion. Monovalent ions like sodium, potassium, and chloride—as well as small divalent ions such as calcium, magnesium, fluoride, and sulfate—are readily removed by RO with high rejection rates of 95-99%. Likewise, dissolved transition metals including, iron, manganese, zinc, and copper, and toxic ions like arsenic, lead, chromium, nickel, and cadmium are rejected based on size constraints.
Organics
A wide range of organic compounds are effectively separated from water by RO membranes. These include man-made organics like pesticides, herbicides, and related agrochemical residues and toxic volatile organic compounds (VOCs) like benzene, toluene, and xylene. Additionally, natural organic matter—including humic and fulvic acids, tannins, and lignins—is removed through size exclusion. In some cases, hydrophobic organics may adsorb strongly to membrane surfaces as well, thereby aiding removal. Rejection rates vary from 90-99%+ based on molecular weight and chemistry.
Particulates
Reverse osmosis readily removes all manner of particulate contaminants like clays, silts, microorganisms, and colloidal particles that would otherwise pass through traditional filters. Typical particulate rejections exceed 99.9%. RO membranes thus act as an absolute barrier, ensuring the complete removal of bacteria, protozoa, viruses, and more.
The Importance of Purification
Reverse osmosis is a vital treatment technology for meeting strict regulatory standards for both drinking water production and wastewater discharge/reuse applications. For drinking water, RO can reliably reduce a wide range of contaminants, including salts, metals, organics, and particulates down to trace levels. This purification process helps achieve the necessary compliance with primary and secondary drinking water quality guidelines.
For wastewater, RO enables effective reuse strategies by providing advanced purification capabilities that allow treated effluent to meet regulatory criteria for either direct discharge into environments or indirect potable reuse after blending with raw water reservoirs.
Process Water Quality Requirements
Across a diverse array of industries, the use of reverse osmosis to purify water is critical for protecting equipment integrity and performance. In sectors ranging from refining, to mining, to textiles, water that’s used for processes like heating/cooling, cleaning, transportation, and reactions must be strictly controlled for parameters like conductivity, silica, organics, and hardness: qualities that impact scale formation, corrosion tendencies and fouling potential. Reverse osmosis reliably removes troublesome dissolved contaminants to produce high-purity water that meets targeted quality metrics for these sensitive industrial operations.
Product Purity Needs
Reverse osmosis also plays an invaluable role in ensuring end product quality and consumer safety across pharmaceutical, food, and beverage production. Water is a critical ingredient, process fluid, and cleaning/sterilization utility in these industries. Any contaminants that are present thus pose risk of dosage form degradation in pharmaceuticals or food/beverage contamination issues. By purifying water down to strict USP or FDA purity levels for use, reverse osmosis protects product integrity, safety, and public health.
Energy Consumption
Reverse osmosis is an energy-intensive, pressure-driven process. Pumping feed water through the membrane to overcome osmotic pressure and achieve desired recovery consumes significant power. Energy use scales with salinity, as higher total dissolved solids (TDS) means higher osmotic pressure requirements. Seawater RO requires up to 10 times the energy of brackish water RO. Brackish water RO uses between 0.2 and 0.4 kWh per cubic meter, while seawater RO uses 2.5 to 4 kWh per cubic meter treated.
Strategies to Optimize Energy Efficiency in RO Systems
There are several ways to optimize the design of reverse osmosis systems for efficiency. Choosing the right membrane and equipment components to balance water recovery and energy use based on feed water conditions and product requirements is key. An expert can evaluate factors like salinity, flow rate, target purity levels and help design a right-sized system that avoids issues with fouling, scaling or shortened membrane life.
Components like energy recovery devices and high-efficiency pumps and motors help reduce energy demand. But ultimately the system must be deliberately engineered for the specific application. Simply buying off-the-shelf reverse osmosis equipment is unlikely to provide an optimized solution. Our team can assess your needs and layout design considerations for an energy efficient system that fits your operational constraints.
Learn more about our water efficiency services and free tools here.
While energy-intensive, continued advancements towards more efficient membranes and system optimization—along with expanding applications in wastewater reuse and seawater desalination—will cement reverse osmosis’s vital role in sustainably meeting freshwater demand across sectors. With unmatched purification power, reverse osmosis will remain indispensable for treating challenging feed waters and producing ultrapure water far into the future.
Reach out to our team today if you need any further consultation on your reverse osmosis system and optimization.